How Life Science Cleanrooms Comply with GMP Regulations: 6 Essential Practices
GMP Cleanroom Compliance: What You Need to Know
Life science cleanrooms are critical environments where lifesaving therapies and drug products are manufactured, tested, and packaged. To ensure the safety, identity, strength, purity, and quality of these products, cleanrooms must comply with Good Manufacturing Practice (GMP) regulations. GMP guidelines—enforced by agencies like the FDA (U.S.) and EMA (Europe)—set rigorous standards that minimize risks in pharmaceutical manufacturing.
Below are six essential ways well-designed cleanrooms help organizations maintain GMP cleanroom compliance.
The 6 Essentials for GMP Cleanroom Compliance
Achieving GMP compliance requires attention to detail across multiple areas. Below are six core standards that define a compliant cleanroom operation.
1. Environmental Control and Monitoring
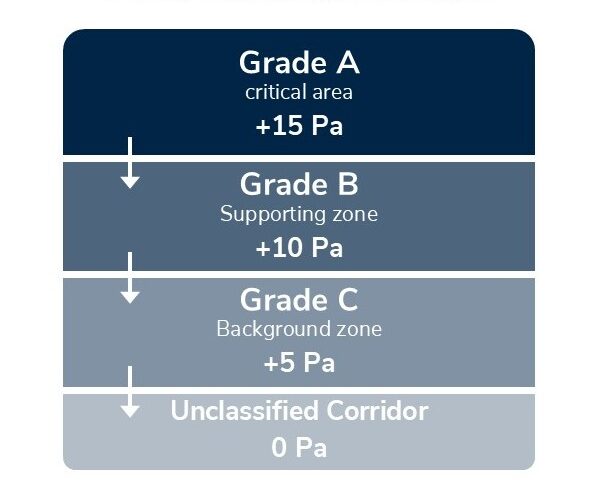
Strict environmental control is the foundation of GMP cleanroom compliance. This includes particle levels, temperature, humidity, and pressure differentials. Cleanrooms are often classified by ISO standards (ISO 14644-1) based on permissible airborne particle levels. For example, an ISO 5 cleanroom allows no more than 3,520 particles per cubic meter ≥0.5μm.
To comply, facilities must:
- Use HEPA filters to remove airborne contaminants.
- Maintain positive pressure to prevent contaminated air from entering.
- Monitor temperature and humidity within validated ranges.
- Perform continuous or frequent particle monitoring.
Any deviation triggers alarms and corrective action to preserve product integrity.
2. Validated Cleaning and Disinfection Procedures
Cleanliness is paramount in pharmaceutical production. GMP mandates that cleaning and disinfection procedures be validated—proven effective and repeatable.
Facilities must ensure:
- Use of GMP-approved detergents and disinfectants.
- Routine sanitization to control microbial contamination.
- Cleaning based on SOPs (Standard Operating Procedures).
- Ongoing microbial testing (swabs, contact plates, air sampling).
Contamination events require investigation and CAPA to maintain compliance.
3. Personnel Hygiene and Training
Human operators are the highest contamination risk. GMP emphasizes strict training, gowning, and hygiene protocols.
Personnel must:
- Wear appropriate gowning attire (coveralls, gloves, masks, shoe covers).
- Receive ongoing GMP and cleanroom behavior training.
- Follow strict entry/exit protocols via gowning rooms or air showers.
- Maintain hygiene standards such as handwashing and no jewelry.
Routine audits, retraining, and qualification ensure adherence.
4. Controlled Material and Equipment Flow
To prevent cross-contamination, GMP requires controlled movement of materials, personnel, and equipment
Key practices include:
- Segregated material airlocks for raw materials, finished products, and waste.
- Sterilization protocols for tools and components.
- Unidirectional workflows for cleanroom activities.
- Dedicated equipment for specific processes when necessary.
These controls safeguard cleanroom integrity and product quality.
5. Documentation and Record Keeping
A central GMP principle: “If it isn’t documented, it didn’t happen.” Comprehensive records ensure traceability and accountability.
Required documentation includes:
- Batch records of each production step.
- Cleaning and sanitization logs.
- Environmental monitoring data.
- Deviation reports and CAPAs.
Regulatory inspections frequently focus on verifying documentation accuracy and consistency.

6. Routine Audits and Continuous Improvement
Compliance is an ongoing commitment, not a one-time effort. GMP requires proactive internal audits, inspections, and quality reviews.
Best practices include:
- Self-inspections to identify deficiencies.
- Third-party audits for objective evaluation.
- Trend analysis of environmental data.
- Implementation of continuous improvement programs.
This cycle of vigilance fosters a culture of quality and compliance.
Real-World Example: RoslinCT’s GMP Cleanroom Suite
Our UK partner Connect 2 Cleanrooms completed a state-of-the-art GMP-compliant facility for RoslinCT, a cell and gene therapy leader.
The cleanroom suite included:
- 3 Grade B cleanrooms
- 2 Grade C cleanrooms
- 15 corridors, lobbies, and changing areas (Grades B, C, D)
- Sample and packing area
- Cryostorage and vaporized hydrogen peroxide (VHP) staging area
- Training laboratory
- Waste room, warehouse, and plant room
- Stairwells, foyers, and bathrooms
- Offices and conference rooms
The large facility obtained full GMP qualification and now operates according to both US and EU GMP standards — ensuring consistent, high-quality results in the rapidly growing, life-saving field of cell and gene therapy.
If you’d like to learn more about this GMP cleanroom project, you can find the full project description here.
Build with Confidence: Partner with Angstrom Life Sciences
GMP cleanroom compliance is essential for safe, effective pharmaceutical manufacturing. From environmental controls to audits, every element of cleanroom design and operation supports patient safety and product quality.
At Angstrom, we design and build GMP-ready cleanrooms that exceed regulatory expectations. From layout planning to integrated monitoring systems, we help ensure compliance from day one.
Ready to upgrade or build your GMP-compliant cleanroom? Contact our Life Sciences team today.