How to Replace Aerospace Cleanroom Filters
Depending on your unique application and ISO classification requirements, your cleanroom filters’ maintenance schedule and routine may look a bit different than others’. In this article, let’s take a look specifically at when and how to replace aerospace cleanroom filters.
What Are Aerospace Cleanroom Filters?
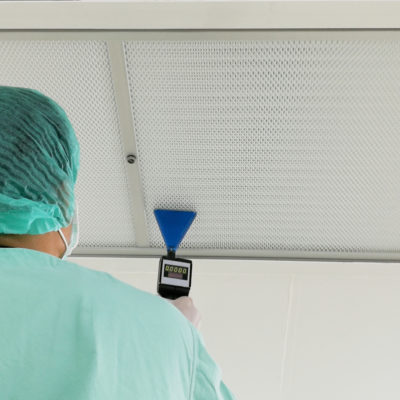
Aerospace cleanroom filters are used to effectively and efficiently maintain air quality throughout the space, removing various hydrocarbon, silicone, and microbiological contaminants. In most cases, these filters consist of three components: a fan, a pre-filter, and a HEPA or ULPA fan filter unit (FFU) — which all work together to treat the cleanroom so it continuously meets a certain cleanliness level, or ISO classification. There are usually many filters that work together to form a comprehensive filtration system, which is installed in the ceiling grid of the cleanroom.
Most aerospace cleanrooms require an ISO classification of 7. This means that the maximum allowed number of particles 0.5µm or larger is 352,000. To achieve this, the cleanroom must have 15-20% of ceiling coverage dedicated to filtration and an average air change rate of about 60-90 completed changes per hour.
However, for some highly sensitive applications like spacecraft hardware and fine electronics, this classification could be lower and more stringent, requiring even more ceiling coverage dedicated to filtration and even faster air change rates.
How to Replace Aerospace Cleanroom Filters
Regularly inspecting, maintaining, and replacing your aerospace cleanroom’s filters is important in ensuring your cleanroom meets those required levels of cleanliness. If left unchecked and dysfunctional, both your products and your end consumers could suffer dangerous and costly consequences. Here’s how to access and replace aerospace cleanroom filters to ensure optimized, safe operations within your facility.
Where to Access Aerospace Cleanroom Filters
As mentioned previously, most filters are housed in your cleanroom’s ceiling grid. However, many cleanroom manufacturers (like us at Angstrom Technology) want to make accessing and replacing cleanroom filters as simple as possible — and climbing up a ladder to breach the ceiling isn’t all that easy.
Therefore, these manufacturers will instead separate the prefilter from the FFU housing and place it in a low wall grille near the floor where it’s more accessible. This way, the ceiling doesn’t need to be regularly accessed and the cleanroom doesn’t need to be recertified with each prefilter replacement.
How Often Aerospace Cleanroom Filters Should Be Replaced
Each part of your aerospace cleanroom filter requires maintenance and replacement at different times.
Pre-filters are the outermost filters, and are therefore exposed to the most contaminants. On average, they’ll need to be replaced six times per year. It’s important to stick to this schedule so that your innermost (and most expensive) filter, the HEPA/ULPA filter, is kept safe for longer periods of time.
Since they are the innermost filters, HEPA and ULPA filters don’t need to be replaced as often as pre-filters. With proper prefilter maintenance, they usually have a lifespan between 7-10 years. However, it’s important to check them more regularly than that to ensure they are in fact functioning properly.
It’s also important to note that your aerospace cleanroom’s location can affect how often your filters need to be replaced. If your facility is located in an area that experiences more air pollution or contaminants (e.g. next to a railroad track or factory), your cleanroom filters may require more frequent inspections and replacements.
Trust a Professional to Replace Your Aerospace Cleanroom Filters
Hiring a professional to inspect, service, and replace your aerospace cleanroom filters can bring many benefits to your facility. They’ll have the knowledge and experience needed to ensure your equipment meets stringent ISO standards, and they’ll help you maintain a consistent maintenance schedule — leaving you with a few less things to worry about.
Looking for someone to service your aerospace cleanroom and get it back in top shape? The experts at Angstrom Technology can help! We’d be happy to put you on our schedule for filter replacement, HVAC upgrades, and more. Contact us for more information.